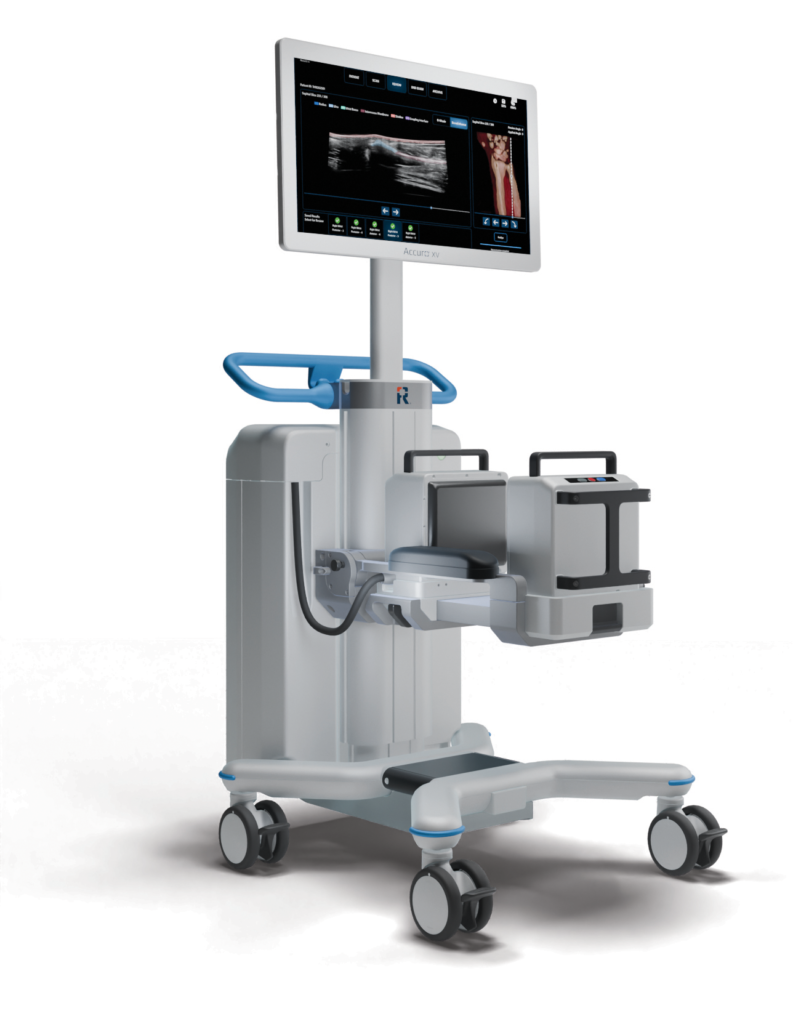
RIVANNA, developer of AI-enabled clinical decision-support solutions, recently announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the Accuro XV Diagnostic Ultrasound System.
“The clearance marks a pivotal regulatory milestone for RIVANNA, authorizing commercial use of the Accuro XV for musculoskeletal imaging by qualified and trained healthcare professionals in hospital and medical clinic environments,” the press release states.
The 510(k) clearance comes as RIVANNA accelerates development of AI-enabled capabilities for the Accuro XV platform, including BoneEnhance, an image segmentation module for enhanced bone visualization, and CADe/x, the first computer-aided detection algorithm for automated fracture identification derived from volumetric ultrasound imaging. Both capabilities are being trained and validated through an ongoing multi-site clinical study spanning eight academic medical centers nationwide.
“FDA clearance of the Accuro XV marks a major inflection point for RIVANNA,” said Will Mauldin, Ph.D., co-founder and CEO. “This clearance demonstrates the scalability of our core platform – shared AI architecture, imaging hardware and regulatory pathway – across distinct clinical applications. With no targeted point-of-care solution in this space, we are positioned to define the standard of care for extremity injury triage.”
The Accuro XV features a conformable three-dimensional volumetric ultrasound probe that automates large field-of-view image acquisition following initial patient positioning, the release states.
“The system’s motorized linear translation technology acquires B-mode images, a 10 cm scan extent; a compliant polyurethane stand-off medium creates a conformable patient contact surface to improve acoustic coupling with irregular anatomies. The cart-based system includes a touchscreen interface, an integrated battery pack for point-of-care mobility, and DICOM-compatible image archival workflows,” it adds.
“With FDA clearance in hand, we can accelerate building the AI capabilities that will unlock the system’s full diagnostic potential,” said Delphine Le Roux, Ph.D., PMP, senior director, market access and strategic partnerships at RIVANNA. “The combination of an automated acquisition platform and AI-enabled fracture detection has the potential to democratize access to high-quality musculoskeletal imaging and reduce the burden on imaging resources in high-volume emergency settings.”
The Accuro XV clearance follows the recent publication of a feasibility study demonstrating that non-physician operators can acquire diagnostic-quality extremity images following just one hour of hands-on training, with image quality comparable to that achieved by board-certified emergency physicians. The study, conducted at UVA Health and UT Southwestern Medical Center and published in the Journal of Emergency Medicine, enrolled 205 patients and found that more than 90% of scans in both operator groups were rated adequate for diagnostic interpretation.




