The Right to Repair Medical Devices: Recapping the Debate

When you buy something, should you have the freedom to get it fixed any way you choose? It’s a question surrounding everything from cars and cell phones to gaming systems and life-saving medical devices.
The “right to repair” movement is making headlines, and the medical imaging community is a part of the ongoing debate. It’s been an issue for quite some time, but discussions over how much control manufacturers should have over repairability gained steam when the coronavirus pandemic hit the United States.
As hospitals saw the first influx of COVID-19 patients, ventilators quickly became critical devices for care. However, some healthcare providers found themselves with ventilators that needed repair and no way to do the work to fix them on their own. That’s because certain original equipment manufacturers (OEMs) did not allow it or do not make certain parts and tools publicly available.
A recent New York Times story on the right to repair movement cites Leticia Reynolds, a medical equipment technician in Colorado. She explained that waiting on a manufacturer during a health crisis is a serious problem.
“‘We’re 100-percent solely depending on the manufacturer,’ she said, even for routine maintenance. But that can delay repairs, which can mean ‘equipment isn’t available for a patient that needs it.’”
From a big picture standpoint, a July 2020 report by the U.S. Public Interest Research Group (U.S. PIRG) found nearly half of the biomedical professionals surveyed said they’d been denied access to “critical repair information, parts, or service keys” since March.
Concerns like this led to new federal legislation over the summer. Senator Ron Wyden (D-Oregon) and congresswoman Yvette Clark (D-N.Y.) introduced what’s known as the Critical Medical Infrastructure Right to Repair Act. The bill’s sponsors are now urging Congress to include the act as part of the next COVID-19 relief package.
What is The Critical Medical Infrastructure Right to Repair Act?
Wyden and Clarke’s bill proposes that trained experts (outside of the manufacturer’s organization) should be able to repair and maintain essential medical equipment during a crisis. That includes having access to the documentation and tools needed to complete the job.
Specifics of the bill include:
-
- Protecting equipment owners and servicers from liability under federal copyright law.
- Allowing for the fabrication of patented parts on a non-commercial basis as needed for repair/maintenance.
- Requiring manufacturers (on reasonable terms) to provide access to tools and information used to diagnose, service, maintain, and repair equipment.
The legislation also calls on the Federal Trade Commission (FTC) to evaluate the bill’s impact on innovation and competition within the critical medical infrastructure market.
When the act was announced in August, Senator Wyden said allowing people with the skills to fix and maintain lifesaving devices is perfectly reasonable, especially during a pandemic:
“It is just common sense to say that qualified technicians should be allowed to make emergency repairs or do preventative maintenance, and not have their hands tied by overly restrictive contracts and copyright laws, until this crisis is over.”
More recently, Wyden co-wrote an opinion piece for Slate with a clinical/biomedical technician. The article brings up the talent shortage in the medical community, including its impact on rural areas. As an example, the authors point to a medical system in Colorado and Kansas where the only certified technician in the area was unavailable for weeks after being exposed to COVID-19.
Alan Morgan, CEO of the National Rural Health Association, raised issues like this in a statement supporting the new legislation:
“As COVID-19 surges across rural America, rural providers must have the rapid ability to maintain effective and operational equipment. This common-sense approach will enable rural providers caring for COVID patients to keep lifesaving equipment operating during this pandemic.”
More on these topics from Technical Prospects:
It’s worth noting that, on a state level, the right to repair tends to cross party lines. There is legislation in 20 different statehouses that has been introduced by Republicans and Democrats alike.
Medical Imaging’s Role in the COVID-19 Pandemic
While ventilators received much of the attention in terms of critical devices for addressing COVID-19, imaging equipment is also playing a part in the pandemic response. You’ll often see medical imaging systems such as CT scans and X-ray equipment mentioned alongside ventilators when the right to repair is discussed.
That’s because healthcare providers are using CT scans and X-ray machines to help properly diagnose and manage cases of COVID-19.
An article from Medscape explains how certain abnormalities in CT scans of patients’ chests are common among those with the virus. CT scanning is also helpful for monitoring and managing patients with COVID-19-related pneumonia.
Mobile X-ray equipment and parts, in particular, are playing a crucial role in diagnosing patients while ensuring only those who truly need to be hospitalized are admitted to healthcare facilities. Clinical Imaging reported on their effectiveness and efficiency:
“The use of mobile x-ray equipment could represent a safe approach, enabling imaging of suspected or confirmed COVID-19 patients, performing examinations in their house or nursing home, reducing social contacts. Of note, it has been already shown that x-ray examinations performed at home (or in the nursing home) using modern mobile equipment provide an image quality at least comparable to those obtained at [a] hospital.”
The flowchart graphic below illustrates how a team would use mobile X-ray systems to safely diagnose patients who may need to isolate at home:
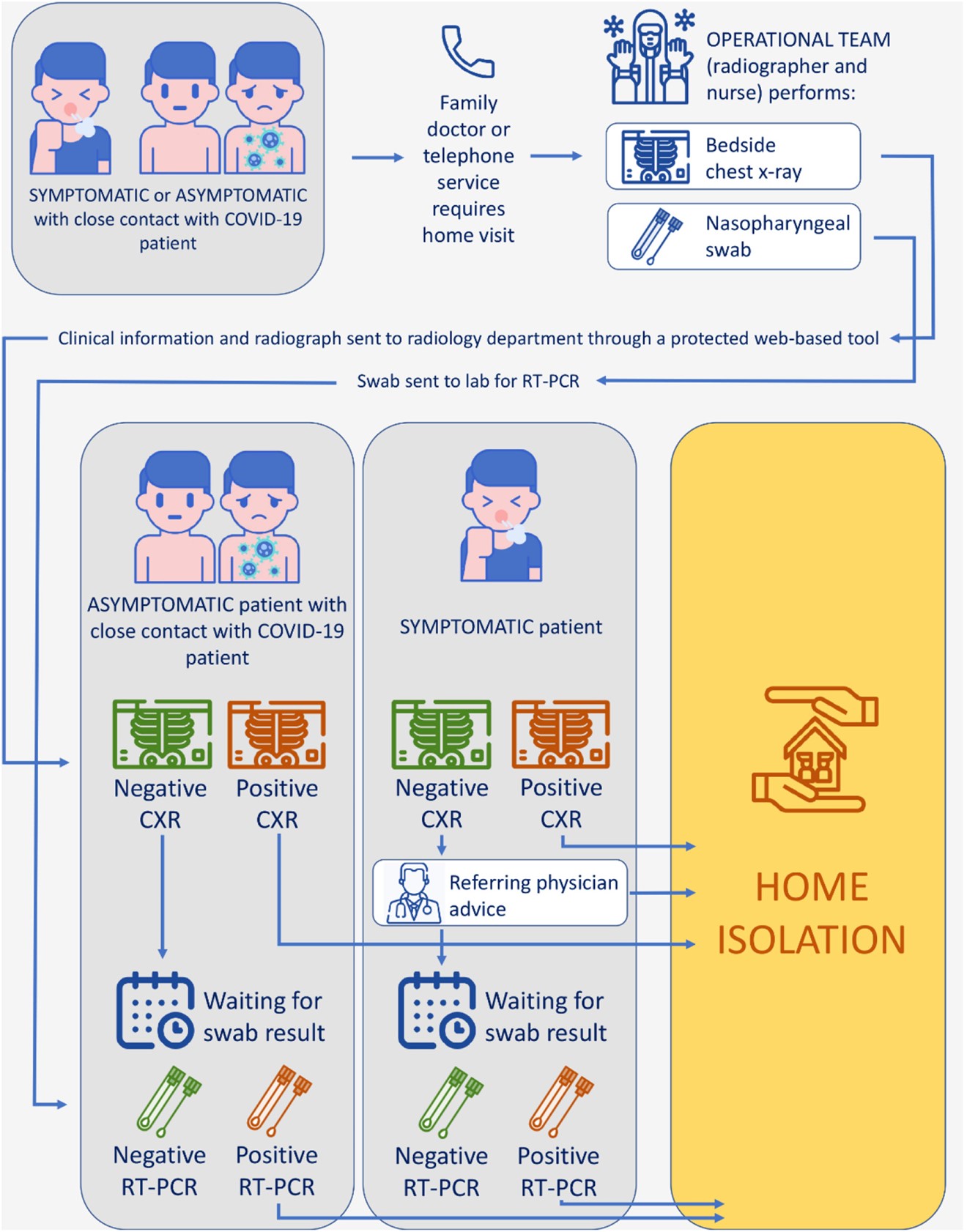
Image credit: Clinical Imaging
As the pandemic continues, keeping mobile imaging equipment operational is extremely helpful. It reduces COVID-19 exposure among frontline healthcare workers as well as the overall spread of the virus because it keeps those who are potentially infected in their homes.
Different Views on Patient Safety
A wide range of healthcare and engineering groups have endorsed the Critical Medical Infrastructure Right to Repair Act, but there are also many opponents of the bill.
When it comes to medical imaging, the different sides of the debate will be familiar to anyone who’s followed the fight over introducing regulation and FDA oversight into third-party organizations such as parts providers and independent service organizations (ISOs). Manufacturers and the organizations that represent them are on one side, third-party organizations are on the other, and healthcare providers tend to fall somewhere in the middle.
All three sides bring up patient safety as the key area of concern, but what that actually means depends on who you ask and what point they’re trying to make.
The Medical Imaging and Technology Alliance (MITA) is one of the more vocal opponents of Wyden and Clarke’s right to repair legislation. In an opinion article for Fierce Healthcare, Acertara CEO and MITA chair G. Wayne Moore claimed manufacturers actually have taken steps to provide more access during the pandemic – specifically for ventilators. However, he believes unfettered access to imaging equipment is problematic, partly because of the wide range of complexity among devices.
“With an unsuccessful or botched repair on an ECG Holter, one can throw it away at relatively minimal risk or expense. Not so with a multimillion-dollar 3T MRI system where a mishandled repair can lead to a quenched magnet, or worse yet, an injured patient.”
Interestingly, a Wired.com article features both sides using cell phone analogies to explain their reasoning around whether or not the right to repair imaging systems makes sense.
Peter Weem of MITA is quoted saying:
“With other goods, if something like a cell phone is improperly repaired and then it fails to perform, the worst-case scenario is that you have to replace the device. Whereas if a medical device is improperly repaired, there’s the risk of injury to the patient or the operator, or death.”
However, Nadar Hammoud, a biomedical engineering manager in California, uses a similar analogy to explain why the ability to quickly and affordably repair a medical device is critical to patient health and safety:
“If the iPhone isn’t fixed, you’re not going to have a phone. If you don’t fix a vent, the patient is dead.”
What’s clear is that no matter which side you’re on, there is the undeniable fact that money is also a factor. For ISOs, increased access to tools, information, and service keys means field service engineers can take on more work without the usual roadblocks. But manufacturers have a vested interest in keeping tools and information proprietary, especially when equipment is under contract.
That doesn’t always create the best scenario for healthcare providers dealing with budget issues because of COVID-19. Hammoud told Wired about an instance in which he could have ordered a replacement part for $80 and fixed a device in-house. But, the OEM required one of its engineers for the job – at a cost of $4,000.
Biomedical engineering director Paul Kelley told Wired the situation is getting worse.
“It’s getting more and more frustrating. We can do less and less work on equipment. We’re getting less and less documentation. Training is getting harder, and parts are getting scarcer.”
Finding Middle Ground on the Right to Repair

Despite any underlying intentions for our respective industries, we can all agree that the health and safety of patients really should be the top priority. If we can all focus on that common ground, we will be able to come to a compromise in this debate.
At Technical Prospects, we believe it’s possible to have both freedom and responsibility when it comes to the right to repair medical imaging systems. Healthcare providers should have the opportunity to fix their own equipment if they employ qualified people. They should also have the option to use reliable ISOs, which could create healthy competition with OEMs. This could make the cost of repairing and maintaining crucial equipment more effective affordable.
Technical Prospects wholeheartedly agrees that proper training is needed. And, it may indeed be necessary to adopt quality management principles across the industry. In the meantime, we’re doing everything in our power to serve the healthcare community.
Technical Prospects focuses on Siemens medical imaging equipment in order to provide the utmost expertise to the engineers we support with reliable replacement parts, those who come to us for training or technical support, and the healthcare providers who use us for service and repairs. Technical Prospects is also ISO 9001: 2015 certified to prove our commitment to quality in every aspect of our work.
During times of crisis such as this, it’s important to have partners you can trust, and that’s what we constantly strive to be for our customers and trainees.




