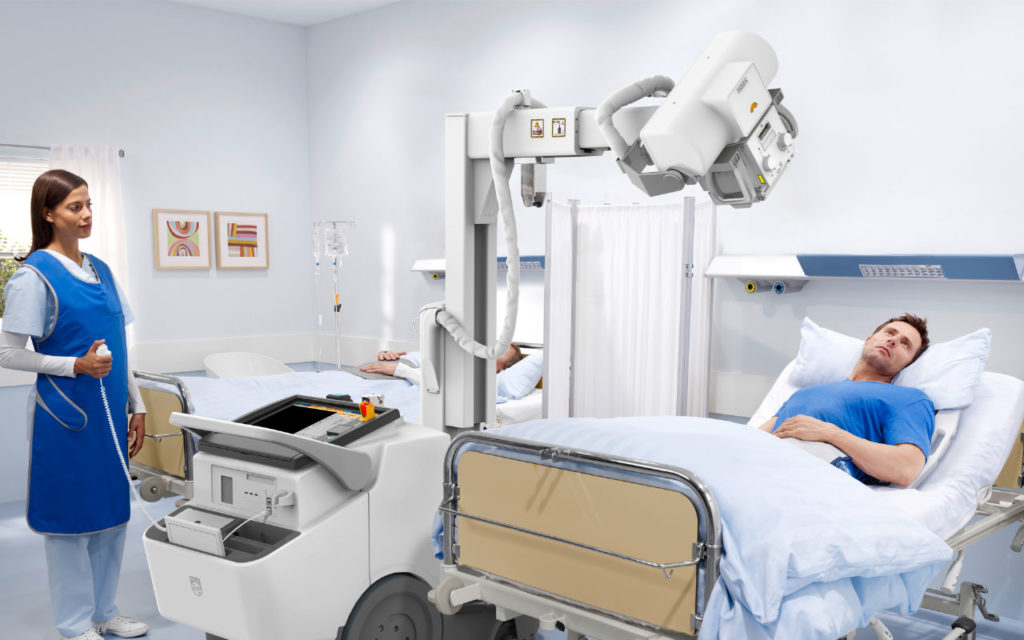
Launched at RSNA 2019, Philips’ MobileDiagnost wDR 2.2 is a premium mobile digital radiography system that brings the superb image quality and high efficiency of its premium mounted systems to a mobile system. The MobileDiagnost wDR 2.2 includes Philips’ Eleva user interface, an easy-to-learn and customizable platform commonly found across a range of Philips digital radiography systems, which helps enable a smooth, efficient and patient-focused workflow. The system also includes a range of security and access management features, workflow enhancements and Philips’ UNIQUE 2 image processing, which provides high-quality images of all anatomical areas.




