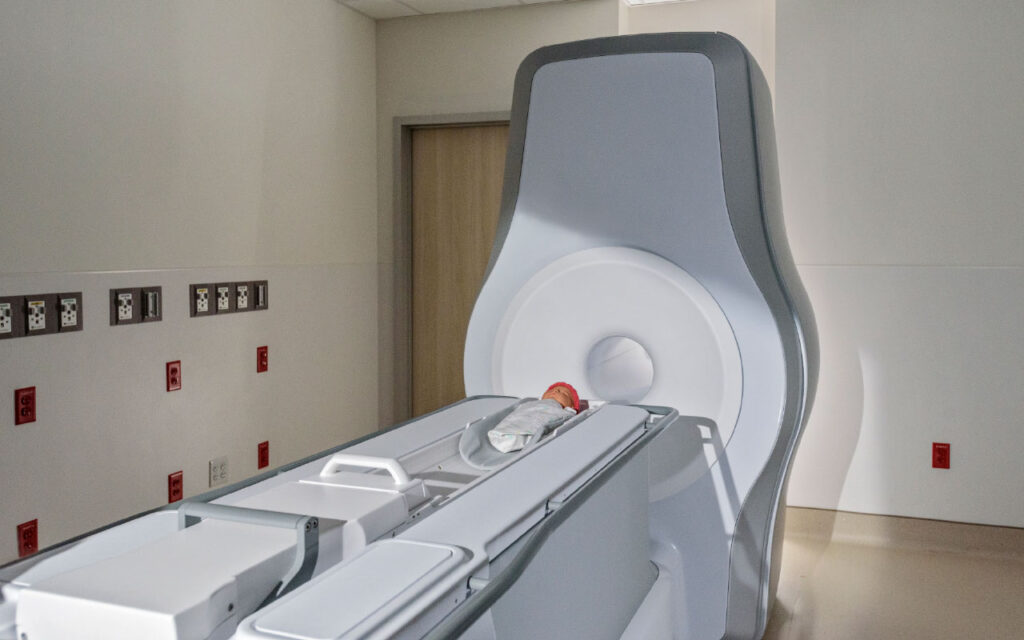
Eyas Medical Imaging Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Ascent3T Neonatal Magnetic Resonance Imaging (MRI) System, a whole-body magnetic resonance scanner designed and optimized specifically for neonate and infant anatomy, including head, body and extremities. The Ascent3T is the first high-field, 3 Tesla (3T) dedicated neonatal MRI system in the world, according to a press release. It utilizes a 3T magnet, enabling a more comprehensive and precise diagnostic tool that provides healthcare professionals with detailed imaging of vital anatomy, including the brain, lungs, heart and abdomen. The Ascent3T addresses the technical limitations of using an adult-size MRI system to image babies and provides clinicians with improved ability to visualize and diagnose disease in the neonatal patient population.




