By Matt Skoufalos
Since the earliest medical applications harnessing the power of X-rays, the diagnostic imaging space has ever expanded with the emergence of new technologies, new applications of existing technologies, and the systems and people that support their interoperability.
POCUS
Although point of care ultrasound (POCUS) has been in use at the patient bedside since the 1980s, technological innovations by Butterfly Network Inc. and others have demonstrated a recent shift away from traditional ultrasound construction methods in favor of a lower-cost, smaller form-factor product. Darius Shahida, chief strategy officer and chief business development officer at Butterfly Network Inc., describes the company’s iQ product, now in its third iteration, as a fundamentally new approach to building ultrasound devices.
“Prior technology was predicated upon piezoelectric crystals, which are prohibitively expensive to manufacture,” Shahida said. “Beyond cost, they’re hand-cut and hard-wired to a specific frequency or imaging application. Our technology, Ultrasound-on-Chip, allows us to image the entire body with a single probe at a really disruptive price point.”
“Any of the large incumbents sell a phased array, a linear, and a curvilinear probe; some are for very shallow vascular applications, others are for deep abdominal applications,” Shahida said. “The beauty of Butterfly is that we can do all of that, achieving a frequency range of 1-12 MHz with one single probe. We’re a digital solution in an analog world.”
In addition to its versatility of function, the Butterfly iQ3 is priced like a high-end, personal electronic device, capable of connecting easily with other handheld peripherals. By creating a product that can be carried like a phone and that interacts with one seamlessly, its design emphasizes both portability and ubiquity for practitioners when they need it. The company expects to further evolve the underlying technology that powers the device “nonlinearly,” Shahida said.
“Our architecture lends itself to other form factors that we are building ourselves – a wearable, patch-based form factor – and other architectures that miniaturize our transducer so that it can fit into and power other form factors,” he said. “We will continue to iterate and improve our performance until we reach a point of equivalency or superiority to cart scanners.”
Butterfly also has worked to make its brand top-of-mind for the same reasons. Medical students at the University of California Irvine and Temple University in Philadelphia, Pennsylvania, were given iQ devices at their white coat ceremonies in 2019 and 2021, respectively, because the deans at those universities believed in the utility of their medical students having POCUS technology integrated into their curriculum as well as their clinicals. Shahida likened the choice to Microsoft giving its Office software suite to college students, the better to prepare them for the white-collar workforce and inspire brand loyalty.
“It was very core to our strategy to align ourselves with the next generation of practicing physicians,” he said. “We want the next generation of practitioners to go out practicing medicine with Butterfly devices in their pockets.”
In addition to the digital-device price point, Butterfly is selling “to every single specialty in virtually every setting globally,” Shahida said; a function of the company investing heavily in its supply chain and an omnichannel e-commerce sales strategy that supports broad dispersal of the iQ+ and iQ3 devices.
“Any licensed health care practitioner can go on our website and purchase a device that we will ship to them in just a few business days,” Shahida said. “In about seven clicks, they can check out and have a device in their hands in a matter of days. That allows us to access the fragmented practitioner market; we also have a very large health systems team that allows us to leverage a direct sales force domestically and in key international markets.”
Butterfly has sold more than 150,000 units, and Shahida said the company has its sights set on even greater gains by working to close the access gap for medical imaging in regions that don’t have it.
“One of the most exciting opportunities that we face is that the following two facts are simultaneously true: two-thirds of the world’s population lack access to simple medical imaging, and about 80 percent of diagnostic dilemmas can be addressed using simple medical imaging,” he said. “I do not believe there’s a condition on Earth for which an earlier diagnosis leads to an adverse outcome. The quicker you get that information, the quicker you can make that decision and alter the pathway of care.”
Butterfly also claims to have “the largest cloud-based, de-identified ultrasound image repository on earth,” to their knowledge, with which to train its Artificial Intelligence (AI) software. Last year, the company launched “Butterfly Garden,” its version of the Apple store and “an AI marketplace for imaging applications,” Shahida said. More than 300 companies have applied to use its software development kit to create and deploy AI-powered tools for iQ users, and Butterfly has signed some 15 to 20 partners to date, he said.
Shahida described its platform as enabling users “to access and interact with all sorts of technologies that help with guidance and capturing images.”
“We created a solution that the largest technology creators failed to create, in part because doing so would disrupt their core business,” he said.
PHOTON-COUNTING CT
Meanwhile, one of the largest players in the medical imaging space, Siemens Healthineers, continues to make inroads in finding additional applications for its photon-counting computed tomography (CT) scanners. Matthew Fuld, Ph.D., director of photon-counting CT at Siemens Healthineers North America, said the technology is having an impact on
cardiovascular, neurosurgery, oncology and even cerebrospinal fluid (CSF) leak patients.
The last of these, Fuld said, have seen results of such significance in identifying the venous fistulas that cause spontaneous intracranial hypotension (SIH) where other forms of imaging cannot, that patients have begun to approach the manufacturer, asking where they could locate health systems that have deployed its NAEOTOM Alpha CT scanner.
“This is a topic I’ve never experienced personally in my career, where I’ve seen patients seeking out Siemens Healthineers to find out where this technology is available,” Fuld said. “They want it in more locations because it enables them to find out where their disease is located; once you can see where the leak is, you can treat it.”
Photon-counting CT is an idea that’s been around for decades, but has taken years, significant research, and financial investments to develop a functional and effective technology capable of measuring every single photon. The complexity involved in manufacturing the cadmium telluride crystals at the heart of the photon-counting detector is significant and expensive. In nature, creating the crystals takes thousands of years; in controlled laboratory environments, growing one ampule of cadmium telluride crystal is a 90-day process, “and if you mess it up on day 88, you start over,” Fuld said.
To that end, the company has constructed a second, large-scale crystal-growing factory in Forchheim, Germany, to complement its first, the better to increase downstream adoption of photon-counting CT.
“We’ve started at the absolute highest end of the technology; now we plan to bring that technology down into our portfolio into facilities that are willing to go down one notch or two,” Fuld said. “Eventually, everywhere in the portfolio is the plan.”
Describing photon-counting CT as “ground-breaking” isn’t just Fuld’s language. In October 2021, when the federal U.S. Food and Drug Administration approved NAEOTOM Alpha scanner for widespread use, the agency itself issued a press release that hailed the device as “the first major new technology for computed tomography imaging in nearly a decade.” It enables the improved spatial and contrast resolution that underpins higher-quality image capture and delivers more effective diagnostic results. Three years later, the manufacturer has seen nearly 450 publications on work enabled by the NAEOTOM Alpha since its launch.
“It’s not just people who want the next new, shiny thing, it’s the topic everyone wants to hear about, and wants to find out more to implement,” Fuld said. “We are expanding and exploring towards what we could do to make it more available to more customers. It’s a technology we see across all our install base in the future, and very positive things – new scanners – in the next short-term.”
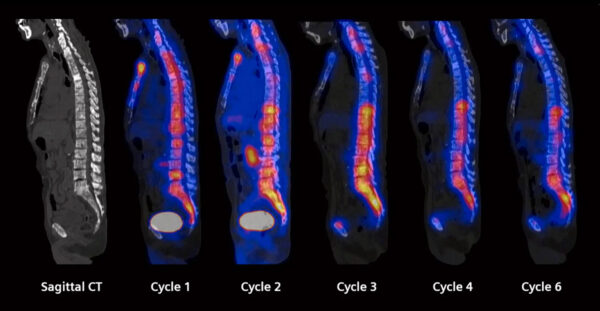
NAEOTOM Alpha users have published studies of patient cases demonstrating “dramatic congenital heart patients with tremendous abnormalities” that have become treatable in ways that they weren’t previously because photon-counting CT provided a pathway for surgical planning.
“Photon-counting CT lets you see all this information at radiation doses that are so low that it would be impractical to attempt that on conventional, energy-integrating CT technology,” Fuld said. “Photon counting is giving people the information to make better diagnostic decisions in many fields. As we have more capability, the cost will come down as we move through.”
THERANOSTICS
For as much as photon-counting CT has made generational advances in CT technology, Siemens Healthineers also foresees increased utilization in theranostics – using one radioactive drug to diagnose disease and a second to deliver targeted therapy – as nuclear medicine and radiation oncology practices begin to customize their own infrastructures from the ground up.
Siemens Healthineers Global Lead of Theranostics Solutions Lady Sawoszczyk said that new technologies can improve workflow and resolve operational issues associated with imaging and radiopharmaceutical manufacturing.
After its 2021 acquisition of Varian Medical Systems, Siemens Healthineers focused on the continued development of the cloud-based Varian ARIA CORE workflow management software, which addresses emerging needs in theranostics. In June 2024, Siemens Healthineers unveiled new theranostics features for the ARIA Systemic Therapy Management (STM) module for ARIA CORE. With drug ordering, pharmacy dispensing, inventory, and administration features, STM is aimed at helping manage cancer patients’ needs and automating tasks to support burned-out staff.
“We see patient volumes going up, and more centers starting to open, but the standard of care for workflow is still being addressed by clinicians and administrative staff,” Sawoszczyk said.
“We found a lot of customers in theranostics are having these challenges, trying to automate a lot of the tasks that cannot be automated. That’s changed quite rapidly, so we’re trying to meet that demand to try to help those institutions.”
ARIA CORE strives to resolve analog, pen-and-paper tasking associated with the regulatory checks related to the process of administering theranostics by automating elements of it. In addition to removing human error from the process, Sawoszczyk hopes that a software-based solution can also close shortfalls in staffing that can also hamper operations.
“As an imaging company providing these scanners to make them more workflow-efficient, we’re also looking at software,” Sawoszczyk said. “I’ve been on that side when I worked in the clinical environment, and we were doing eight infusions a day. If I could find a way to automate what we were doing, I could maybe provide two times more infusion appointment for patients, and maybe do 16 patients a day.”
Another difficulty in the field involves growing the myriad applications for theranostics interventions when the clinical trial process is so arduous and time-consuming. Sawoszczyk spoke about efforts to spur collaboration among researchers, professional imaging specialty societies, and the FDA itself to deliver these life-saving treatments more quickly to patients who need them.
“We want to make sure that we ensure the safety of these theranostics patients as we move from third-line to first-line treatments,” she said. “Novartis just published a very promising trial for first-line treatment of neuroendocrine tumors, and I am hopeful they will submit for FDA approval sometime in 2025.”
“The supply chain will continue to be a challenge,” Sawoszczyk said. “Imaging companies also have been looking to partner with pharmaceutical companies to try to solve some of these challenges. It’s a global challenge, but there are companies that are building massive infrastructure.”
To her point, the billion-dollar, 70,000-square-feet radiopharmaceutical therapy agent manufacturing facility that Novartis opened in Indianapolis is only one of many that’s needed to develop, produce, and distribute the supply of therapeutic agents at the heart of theranostics. Until radiopharmaceutical companies can supply the field, various vendors, physicians, and professional societies will be relied upon to facilitate its future growth, Sawoszczyk said.
“Every physician believes that they are practicing theranostics in one particular way, and that’s the only way to practice theranostics,” she said. “But if you take a physician practicing in the middle of the United States, and a physician practicing in New York, their resources are very different. There’s more than one way to practice theranostics. As long as you’re able to treat the patient effectively, safely, and efficiently, then patients will be able to benefit from theranostics.”
SOFTWARE
Just as meaningful to the medical imaging space as innovative, cutting-edge, or entirely novel diagnostic and therapeutic technologies is the software that underpins the organization, security, and accessibility of their data. Jordan Bazinsky, CEO of medical imaging management company Intelerad Medical Systems, believes that the next phases of imaging innovation will turn on “fully unlocking the value of generative AI and the cloud.”
“In the context of imaging, there’s so much value there,” Bazinsky said. “Scalability of viewing power and storage power is incredible, as is the security structure, and the ability to collocate people who are dispersed regionally in this work from wherever environment.”
“Generative AI can support improvements in workflow on the diagnostic side,” he said. “How do we help radiologists or clinicians identify true positives, boot out false positives, and work on ensuring that we’re putting all these cloud value points out for our customers and making a first-class system?”
Bringing medical imaging technology to the point of care also helps improve accessibility by reducing workflow steps, operational costs, and other barriers to the delivery of care. With about half the global population lacking access to medical imaging technologies, distributed computing and storage networks can help close some of that gap.
“Medical imaging is the tip of the spear,” Bazinsky said. “You can’t treat someone if you can’t diagnose them. There is complex imaging that can only happen in certain settings of care.”
“Much farther out, what’s the transformative thing that is going to happen in medical imaging that’s going to really change the role of imaging in society and in the health care system?” he asked. “Personalized medicine, that’s really exciting; being able to capture more at ever-earlier stages, all in an attempt to intercede much more quickly.”
“To think that ultrasound is only 68 years old, CT only 53, MRI only 47, we remain in such early stages of what’s possible,” Bazinsky said. “The types of interactions in the body that are being looked at doesn’t lessen the need for really talented clinicians; it just shifts how they spend their day.”
Software products like InteleShare are Intelerad’s answer to facilitating the freer, more intelligent, more rapid exchange of medical images among referring physicians, specialists, and even patients themselves. Bazinsky spoke about working to anticipate “the many endpoints that no one could solve for” by managing data more intelligently within physician workflows. AI-powered solutions can support better-quality care, fewer adverse events, and “better understanding of the patient chronology,” i.e., electronic medical records.
“All that data in the past lives inside of paper charts,” he said. “In this day and age, we’re much better equipped to hand that information out, but a clinician still has to sort through that information,” he said. “There’s plenty of data out there with which to train models, but for smaller populations, like pediatrics, the event isn’t quite as large, so it can be a lot harder to identify a disease state.”
“There’s a lot out there waiting to get unlocked,” Bazinsky said. “There’s plenty out there to get better at for most companies, but even when that happens, things will evolve more. I expect that there’s this finish line that doesn’t get reached.”




